To organize the elements, Mendeleev wrote down their chief properties on cards and arranged the cards into groups with similar properties. He then organised groups and the elements wit in the groups, according to their characteristic mass. Other scientists had arranged the elements into groups according to mass; however, Dmitri Mendeleev had three key steps in his organizing of the periodic table.
Dmitri left gaps in the periodic table because he was so sure that he predicted both physical and chemical properties of the missing elements in the table. When he got evidence from his experiments and they were contradictory, he was willing to place elements into different groups. Mendeleev did not worry about placing elements in his table in the wrong order of atomic mass if the common properties supported his theory about groups.
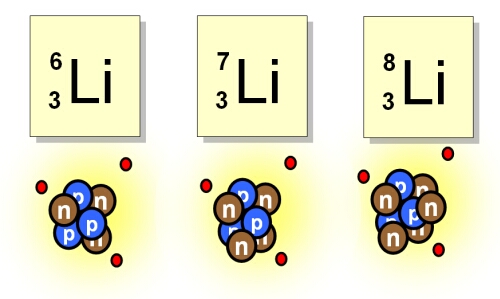
Dmitri back himself so much when predicted there were missing elements, in the end he was right when other scientists found the missing elements and their properties matched those by Mendeleev. The finding of isotopes supported his decision to consider properties before mass in his arrangement.
The periodic table today looks different from Mendeleev's table. Like the elements are now placed in order of atomic number, not atomic mass. Another reason is that there have been more discovery's of new elements, when Dmitri Mendeleev created his table there were 63 known elements; today there are 114 elements. There have been more then 700 versions of the periodic table since the first one was produced since the first one was presented in 1869.
Dmitri left gaps in the periodic table because he was so sure that he predicted both physical and chemical properties of the missing elements in the table. When he got evidence from his experiments and they were contradictory, he was willing to place elements into different groups. Mendeleev did not worry about placing elements in his table in the wrong order of atomic mass if the common properties supported his theory about groups.
Dmitri back himself so much when predicted there were missing elements, in the end he was right when other scientists found the missing elements and their properties matched those by Mendeleev. The finding of isotopes supported his decision to consider properties before mass in his arrangement.
The periodic table today looks different from Mendeleev's table. Like the elements are now placed in order of atomic number, not atomic mass. Another reason is that there have been more discovery's of new elements, when Dmitri Mendeleev created his table there were 63 known elements; today there are 114 elements. There have been more then 700 versions of the periodic table since the first one was produced since the first one was presented in 1869.